Posted Date:- Jul 06, 2021
We are perhaps looking at a future in which industrial waste will form the base for energy storage in batteries. Scientists have shown that the spent catalysts from the energy industry or the raw material for recycling operation that deliver fresh catalysts and valuable metals work as an efficient bifunctional oxygen electrocatalyst and can catalyze the core reactions that facilitate the operation of metal-air batteries.
It can help develop new strategies for effectively using industrial waste for energy storage in batteries paving the way to realize the dream of achieving ‘today’s waste is tomorrow’s energy’.
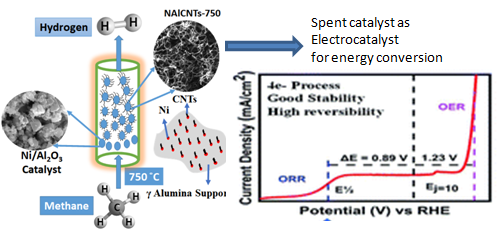
Hydrogen energy offers a promising power generation route for the industry and transport sectors due to high energy density and clean output. One of the ways to produce hydrogen is by catalytic decomposition of methane using nickel catalyst embedded on alumina or zeolite. After several runs, the catalysts get spent due to carbon choking and lose their activity. The spent catalysts are typically subjected to energy-intensive processes such as high-temperature combustion for recycling, releasing a large amount of COx into the atmosphere during the process or chemical treatment for the reclamation of metal constituents. These protocols are neither economically viable nor environmentally friendly, thus demanding alternative avenues to efficiently use the spent catalyst.
One of the best possible routes is to use the retrieved spent catalyst for energy generation/storage applications. The composition of the given spent catalyst, Carbon nanotubes with Ni nanoparticles and porous alumina, may be ideal for direct use as electrocatalyst in electrochemical energy applications and thus, opens up a viable strategy for converting waste to wealth.
Dr C. Sathiskumar, Dr Neena S. John and Dr H.S.S. Ramakrishna Matte from the Centre for Nano and Soft Matter Sciences (CeNS), an autonomous institute under the Department of Science & Technology, Government of India, in collaboration with Hindustan Petroleum Corporation Ltd (HPCL) R&D Green Centre, Bengaluru, have demonstrated that the above-spent catalyst works as an efficient bifunctional oxygen electrocatalyst. It can catalyze both electrochemical oxygen evolution (OER) and oxygen reduction reactions (ORR), the core reactions that facilitate the operation of metal-air batteries.This research was recently published in the journal ‘Sustainable Energy Fuels’.
The spent catalyst shows stable current density for 20 hrs and 8 hrs towards OER and ORR. The potential difference for overall oxygen electrocatalyst (ΔE) reveals a superior bifunctional activity of the spent catalyst. Furthermore, the spent catalyst employed in Zn-air batteries displayed commendable charge-discharge performance up to 45 hrs with high reversibility.
The work supported by the Centre for High Technology (CHT)-Oil and Industry Development Board (OIDB), Hydrogen Corpus Fund help in effectively utilizing industrial waste for energy storage applications, thus producing green energy in a sustainable manner.
Publication links:
DOI: 10.1039/d1se00007a.
For more details Dr Neena S. John and Dr H.S.S. Ramakrishna Matte jsneena@cens.res.in, matte@cens.res.in can be contacted.
Ministry of Science & Technology Press release dated 06th July 2021
SS/RKP